On December 17, 2025, the Official Journal of the European Union published the Implementation Guide No. C/20256721, which released a Q&A interpretation for the Regulation (EU) 2024/3190 (ban on BPA in contact materials). This guide provides explanations for core operational issues such as the scope of application of the BPA ban, the definition of restricted substances, compliance requirements, and transition period in the form of Q&A, aiming to help enterprises smoothly respond to the upcoming comprehensive ban.

Regulatory and supervisory background
Bisphenol A (BPA) is a chemical widely used in the production of polycarbonate plastics and epoxy resins, commonly found in the coating of food cans, plastic containers, etc.
Due to its potential endocrine disrupting properties, the EU has further tightened its use restrictions based on continuous risk assessment, officially passing the RegulationEU) 2024/3190 on December 19, 2024; further strengthening the control, not only implementing a strict ban onPA, but also including other bisphenol substances with specific hazardous properties into the scope of control, on the basis of revising (EU) No 10/211 and repealing (EU) 2018/213.
Following the promulgation of the Administrative Provisions one year ago, the corresponding Q&A guidelines were officially released, providing detailed interpretations of the original. The following is an interpretation of the key points of the guidelines.
Clear points of the Q&A guide
1. Scope of application interpretation
Exclusions from scope: The regulation explicitly excludes from its scope paper and paperboard, recovered materials (due to trace amounts of contamination unintentionally introduced) andamelware.
Definition of scope: In (EU) 2024/3190, it is clarified that it covers materials and articles such as plastics epoxy resin coatings, printing inks, adhesives, etc. In this Q&A Guidance, it is further clarified that external components of food contact materials, they do not directly come into contact with food, but under the conditions of use may result in migration of harmful substances to food, are also subject to the ban. The entire production from raw materials, intermediate products to final products is covered.
2. Define the boundaries of bisphenol management
Comprehensive ban: The use of BPA is strictly prohibited in principle during the manufacturing process, with the exception of two special-use exemptions provided in Annex II of Regulation (EU) 2024/3190. In addition, Article 5 of the Regulation extends the scope of the ban to otherisphenol derivatives classified as CMR (carcinogenic, mutagenic, and reprotoxic) substances in category 1A/1B or as endocr disruptors in category 1, including bisphenol S (CAS: 80-09-1), 4,4'-(1,3dimethylbutyl)bisphenol (CAS: 6807-17-6), phenolphthalein (CAS: 7-09-8), bisphenol AF (CAS: 1478-61-1), and tetrabromobisphen A (CAS: 79-94-7).
Residual requirement: Bisphenol A diglycidyl ether (BADGE) is still listed in EU 10/2011 an allowed plastic monomer but food contact materials and articles manufactured from it need to be ensured to be free of BPA residues.
3. Declarations and tests compliance obligations
Declaration of Compliance and Test Requirements: All food contact materials (including multi-material products) within the scope of regulatory control shall be issued and communicated a Declaration of Compliance (DoC) corresponding to the phase of the operator; at the same time, it can be confirmed whether bisphenol A or bisphenol-class are used based on the information of raw materials transmitted in the supply chain. Although the regulations do not require mandatory testing of materials and products, testing methods are also inevitable options in to prove that there is no residue of BPA and its derivatives in materials or products.
4. Import and export rules
Export requirements: FCM or articles intended to come into contact with food exported from the EU to third countries are not, in principle, subject to this ban but they still need to have full traceability documentation, including clear indication of the destination, so that Member States can check these materials and confirm whether they have been diverted and placed the EU market.
Import requirements: Regardless of where the product is made, materials and articles entering the EU market must comply with the BPA ban requirements, and it is the im who bears the responsibility for compliance verification.
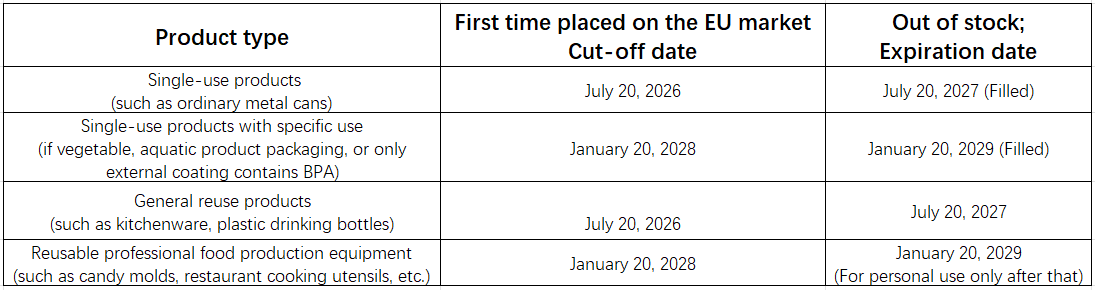
5. Transition buffer period
The Q&A guide contains detailed transition period end dates for different products, summarized as follows: